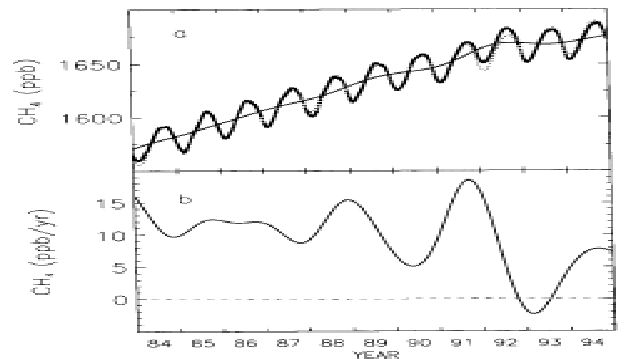
With advanced technology, it is now possible to measure the concentrations of OH directly. It is very hard, however, to construct global OH fields from these local measurements. It is the globally integrated OH that determines the oxidizing capacity of the atmosphere, e.g. the rate at which methane is removed from the atmosphere. For future predictions it is also important to determine whether global OH is increasing or decreasing. One might try to determine global OH from the methane budget. The principle is simple. The amount of methane that is removed by OH is balanced by the emissions and the change in the atmospheric burden of methane. The burden of atmospheric methane can be determined by measurements at a number of remote stations (e.g. the NOAA network). These measurements reveal that the atmospheric burden is generally increasing, but with a variable rate (see figure d).

Figure d: Global average methane concentration (a, parts per billion) and the rate of change (b, parts per billion per year) from 19984 to 1994. The decreased growth rate after 1991 is generally attributed to the eruption of Mt. Pinatubo. This eruption might have influenced tropospheric OH concentrations (e.g. through the photolysis of ozone) but also the emissions of methane from natural wetlands (from Dlugokencky et al., 1996).
The obvious drawback of this budget method is that the uncertainties in the methane emissions are very large. Therefore, the accuracy at which the global OH concentration can be determined is limited. For this reason, scientists have been looking for other compounds that might be used to constrain the global OH concentration, and hence the oxidizing capacity of the atmosphere. Methyl chloroform (1,1,1 trichloro-ethane), a compound that is (almost) exclusively of anthropogenic origin, turned out to be a very useful compound.